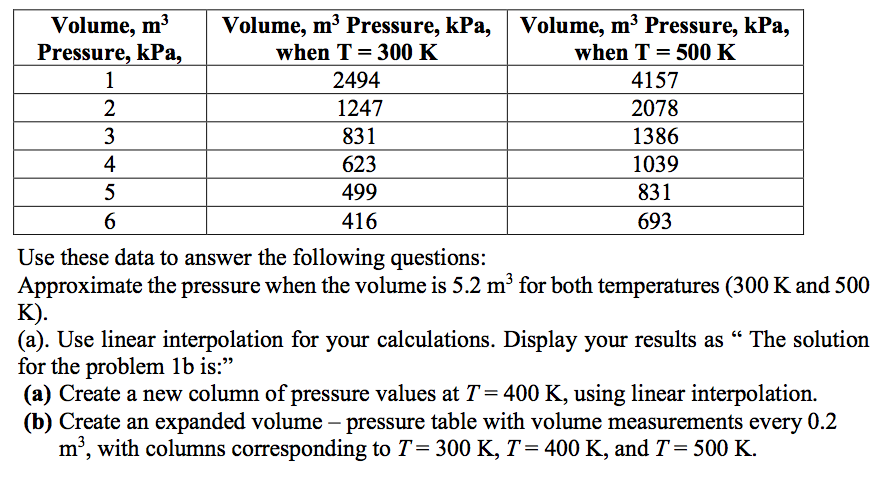
An engine consists of a constant volume process at 0.7 m^3 from 137 kPa to 399 kPa, an isothermal expansion until the pressure drops to its original value, and a constant pressure

Pressure versus groove volume of single-screw expander (p su = 800 kPa,... | Download Scientific Diagram

Air in a piston/cylinder arrangement, shown in the figure below is at 200 kPa, 300 K, with a volume of 0.5 m3. When the piston just touches the stoppers, its volume is
Effect of operating temperature. Pressure, 1013 kPa; reaction volume,... | Download Scientific Diagram

The relationship between the air volume, measured at 4.90 kPa, and the... | Download Scientific Diagram

Volume of effluent needed to increase 10 kPa the filter head loss. The... | Download Scientific Diagram

SOLVED: A bicycle pump contains 0.650 L of air at 101 KPa. If the pump is closed, what pressure is required to change the volume to 0.250 L? 16.41 kPa 262.6 kPa 621.54 kPa 38.85 kPa

SOLVED: What volume would 22.0g of CO2 occupy at a temperature of 25 degrees celsius and a pressure of 105 kPa?
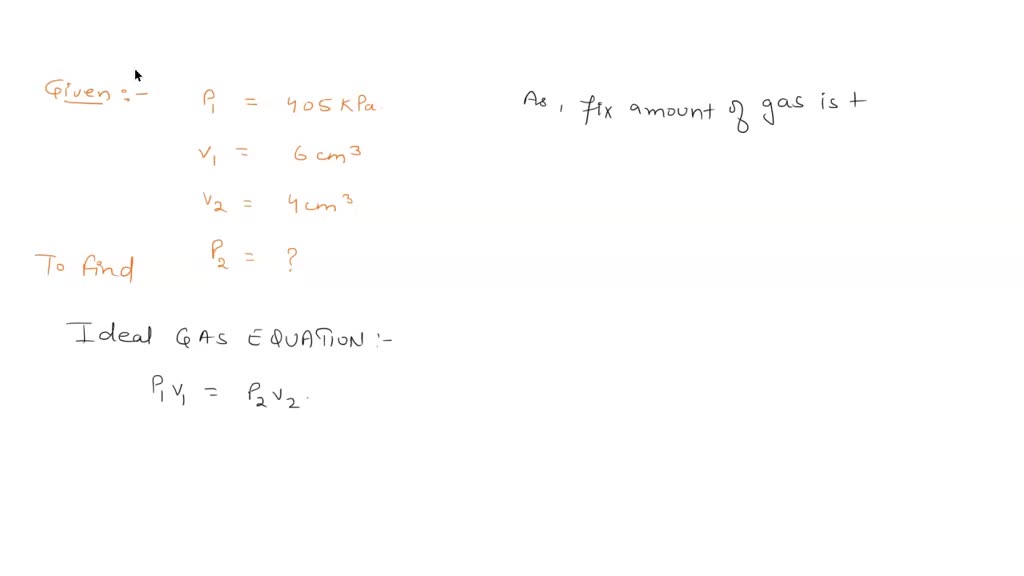
SOLVED: Question 1: At a pressure of 405 kPa, the volume of a gas is 6.00 cm3. Assuming the temperature remains constant, at what pressure will the new volume be 4.00 cm3? (